|
Chlorine – A Great Disinfectant!
There are distinct differences between a Sodium Hypochlorite solution, a Calcium Hypochlorite solution and an onsite generated Hypochlorous Acid solution.
Sodium Hypochlorite Solution (NAOCL)
MSodium Hypochlorite solution often called bleach usually containing LYE is manufactured at a factory, stored, shipped to distribution centers, stored again and then sold.
Calcium Hypochlorite Solution (CAOCL)
Dry Calcium Hypochlorite tablets produce a “FRESH” Hypochloritesolution when mixed with water. In tests done, a solution produced with the proper Calcium Hypochlorite tablet, can maintain “Free Available Chlorine” or Hypochlorous Acid the active disinfectant in this Calcium Hypochlorite solution, for ONLY about 4 hrs, then it starts rapidly degrading.
Hypochlorous Acid Solution (HOCL)
Hypochlorous acid is one of chlorine byproducts that is obtained by dissolving chlorine in water. It is a weak acid. HOCL is the most active ingredient in chlorine solution which its bactericidal effect is 80-100 times more powerful than hypochlorite ion (OCL-). Only 10-30 ppm of HOCL is enough for using as a bactericide. The bactericidal effect of 10ppm of HOCL solution is equivalent to 100ppm of NaOCl solution.
Free available Chlorine content
For chlorine solution to be a good disinfectant it must meet the Chlorine Demand. The chlorine demand is the amount of Free Available Chlorine (FAC) often called Hypochlorous Acid (HOCl), needed to disinfect or oxidize organic matter before a FAC residual is reached. If the chlorine demand is not met then complete disinfection has not been obtained. One of the best signs that the Chlorine Demand has not been met is the strong chlorine odor.
If chlorine solution does not contain enough HOCl to satisfy the chlorine demand of the surface or product to be disinfected, chloramines will form as chlorine and nitrogen-based materials combine. Examples of nitrogen-based materials are proteins and blood. Chloramines are responsible for the obnoxious odor sometimes associated with chlorine disinfection. The obnoxious, pungent, eye-stinging smell of chloramines, mistakenly identified as free chlorine, indicates that the chlorine/water mix is not effective. There is not enough HOCl to satisfy the chlorine demand
Chlorine Efficacy determined by pH
Chlorine in water splits into two forms, Hypochlorous Acid (HOCl) and Hypochlorite Ion (OCl-). How much of each is present in a chlorine solution is totally dependent upon the pH of the solution. As pH rises, less Hypochlorous Acid and more Hypochlorite Ion is in the solution. As the pH rises, less germ killing power is available. According to a University of Illinois study, HOCl is 120 times more effective as a sanitizer than the -OCl ion. The ideal pH of a disinfecting chlorine solution is a pH of 6-7.
Most FRESH Calcium Hypochlorite solutions have a pH between 7 and 8. ALL (fresh or old) Sodium Hypochlorite solutions, (“bleach”) have a pH of 10.25+ producing very little HOCL. These solutions produce mainly the OCl- ion, a very poor disinfectant which is from 80 to 120 times less effective as a disinfectant than HOCl, providing that there is any chlorine left in the stock solution.
Slightly Acidic Hypochlorous Acid
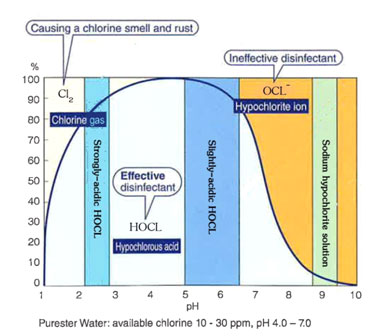
The above chart explains the “Equilibrium chart on chlorine in solution” and also shows how chlorine exists in solution. Three phases of Cl2, HOCL and OCL- are showed in this chart and its composition changes according to pH value. If pH is 4.0 to 7.0, a lot of HOCL will be formed. Since it is proved that the main disinfecting agent is HOCL, hence it can be used to disinfect effectively although the concentration of chlorine is only 10-30 ppm. On the other hand, sodium hypochlorite solution contains only around 5% of hypochlorous acid; therefore a very high concentration of chlorine of 100 to 200 ppm should be used. The rest 95% is OCL- which bactericidal effect is very low, remain unused and will be discarded in wastewater.
Advantages of slightly acidic hypochlorous acid water
- It contains very low concentration of chlorine and is nearly odorless and has no negative impact on foods.
- It is very effective to micro-organisms, even to spores.
- It is very gentle to environment and also to human beings who handle it.
- Wide range of applications, food industry, agriculture, medical field etc.
- It does not contain any toxic substances such as chloroform, chloric acid and bromic acid and very safe.
Reference: George Clifford White Handbook of chlorination and Alternative Disinfectants, 3rd edition, 1999
|